Expo
view channel
view channel
view channel
view channel
view channel
view channel
Medical Imaging
AICritical CareSurgical TechniquesPatient CareHealth IT
Business
Events
Webinars

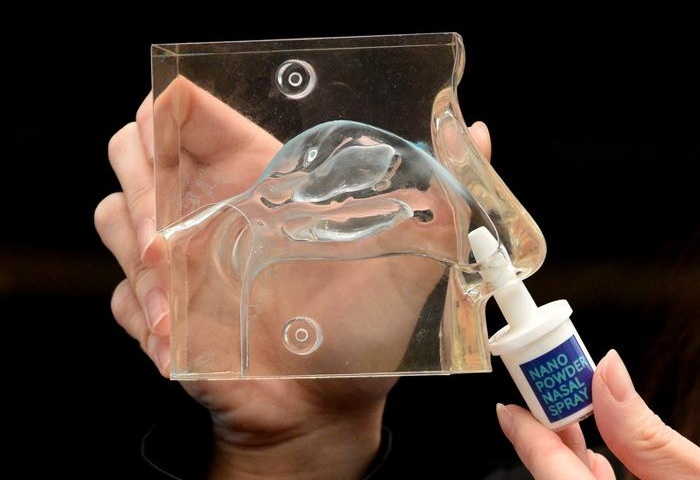
- New Nasal Spray Enables Prehospital Neuroprotection in Ischemic Stroke
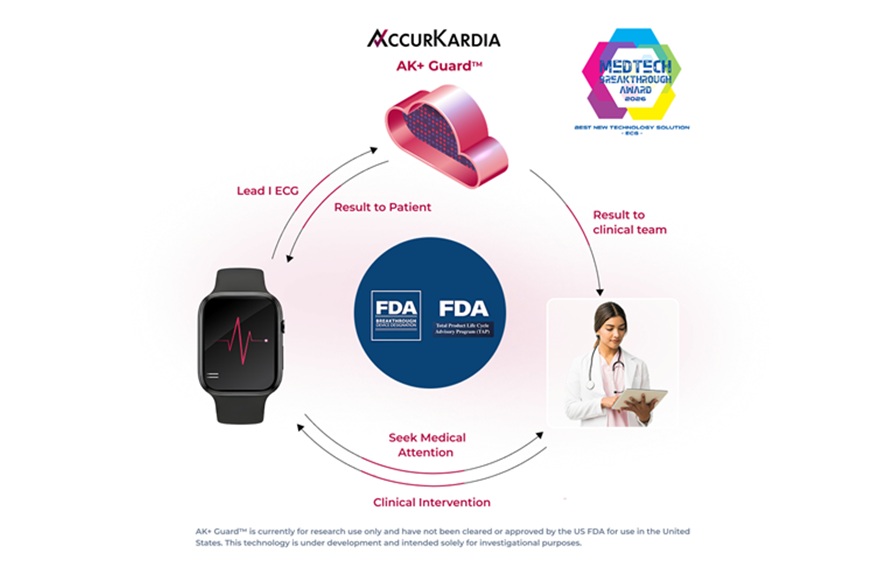
- Single-Lead AI ECG Tool Detects Moderate-to-Severe Hyperkalemia Outside Clinic
- Reduced-Intensity Transplant Regimen Expands Donor Access in Sickle Cell Disease
- AI-Enhanced ECG Screens for Heart Failure Risk in Resource-Limited Settings
- Battery-Free ECG Patch Enables Continuous Arrhythmia Monitoring
- Photoacoustic Imaging System Maps Hidden Nerves and Vessels in Robotic Laparoscopy
- New Endoscopic Device Offers Minimally Invasive Approach to Obesity Management
- Robotic Microsurgery System Gains CE Mark for Clinical Use in Europe
- Robotic Surgical System Enables Gastric Bypass in Space-Constrained Operating Rooms
- Smart Soft Sensors Provide Real-Time Force Feedback in Minimally Invasive Surgery
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
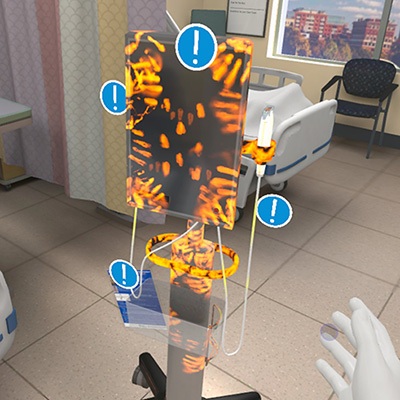
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
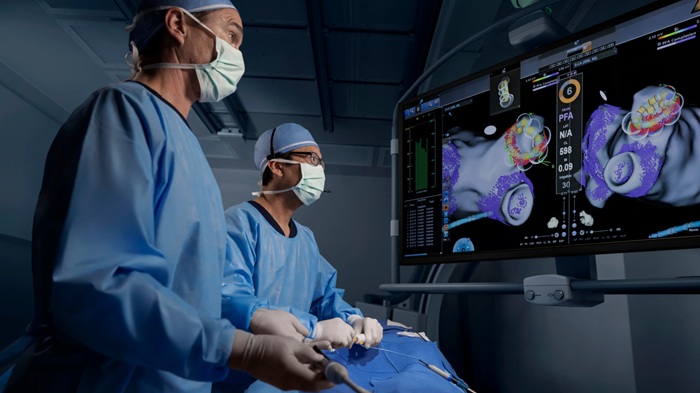
- Johnson & Johnson Launches AI-Driven Cardiac Mapping System
- Proximie Advances AI-Driven Intelligent Operating Rooms with NVIDIA Collaboration
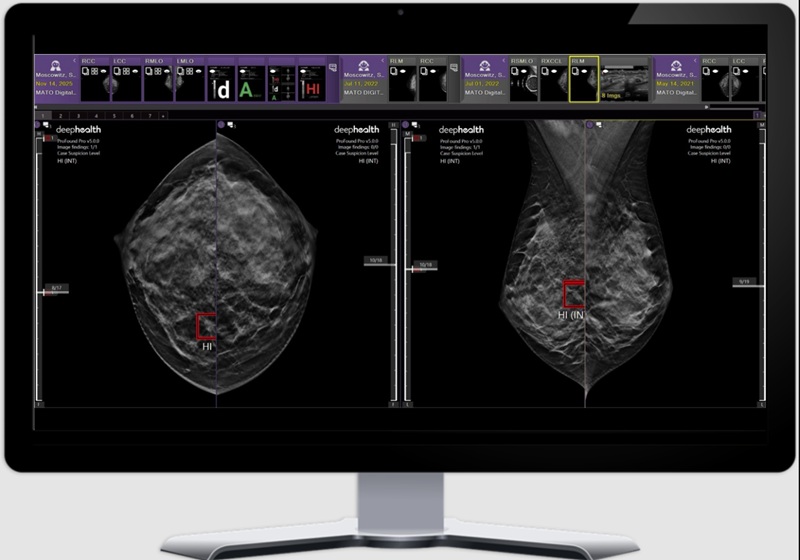
- GE HealthCare, DeepHealth Expand AI Breast Imaging Collaboration
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- New Partnership Advances Physical AI into Perioperative Workflows
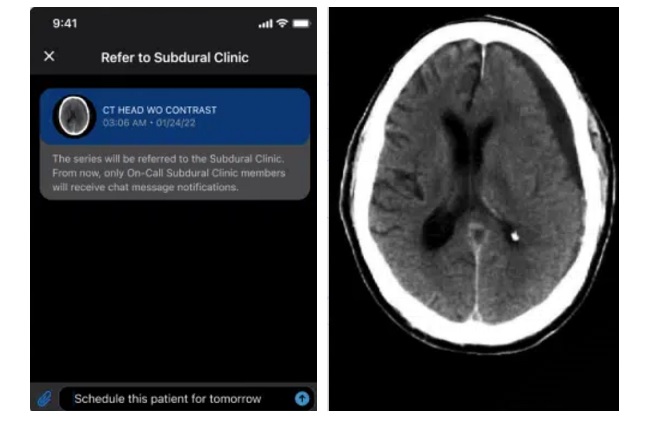
- AI System Detects and Quantifies Chronic Subdural Hematoma
- Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
- Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Facial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
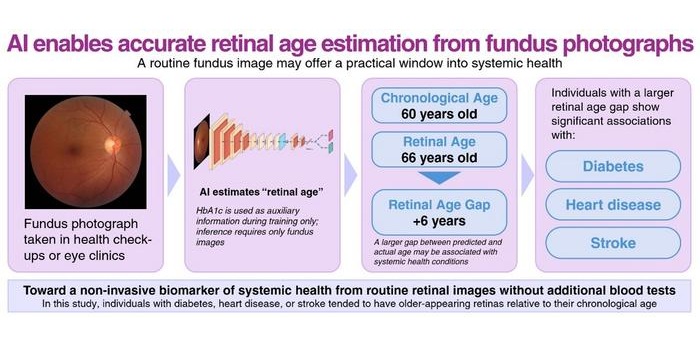
- AI Model Uses Eye Imaging to Identify Risk of Major Systemic Diseases
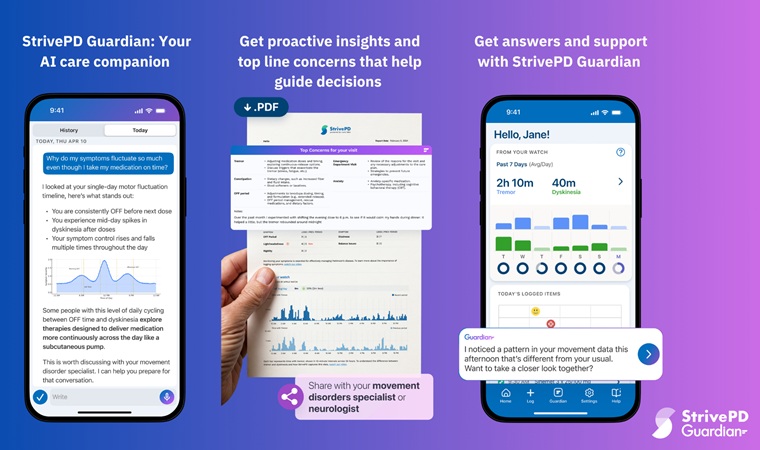
- AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk

 Expo
Expo
- New Nasal Spray Enables Prehospital Neuroprotection in Ischemic Stroke
- Single-Lead AI ECG Tool Detects Moderate-to-Severe Hyperkalemia Outside Clinic
- Reduced-Intensity Transplant Regimen Expands Donor Access in Sickle Cell Disease
- AI-Enhanced ECG Screens for Heart Failure Risk in Resource-Limited Settings
- Battery-Free ECG Patch Enables Continuous Arrhythmia Monitoring
- Photoacoustic Imaging System Maps Hidden Nerves and Vessels in Robotic Laparoscopy
- New Endoscopic Device Offers Minimally Invasive Approach to Obesity Management
- Robotic Microsurgery System Gains CE Mark for Clinical Use in Europe
- Robotic Surgical System Enables Gastric Bypass in Space-Constrained Operating Rooms
- Smart Soft Sensors Provide Real-Time Force Feedback in Minimally Invasive Surgery
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Johnson & Johnson Launches AI-Driven Cardiac Mapping System
- Proximie Advances AI-Driven Intelligent Operating Rooms with NVIDIA Collaboration
- GE HealthCare, DeepHealth Expand AI Breast Imaging Collaboration
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- New Partnership Advances Physical AI into Perioperative Workflows
- AI System Detects and Quantifies Chronic Subdural Hematoma
- Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
- Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Facial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
- AI Model Uses Eye Imaging to Identify Risk of Major Systemic Diseases
- AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk