Expo
view channel
view channel
view channel
view channel
view channel
view channel
Medical Imaging
AI
Surgical TechniquesPatient CareHealth ITPoint of CareBusiness
Events
Webinars

- Noninvasive Skull Sensor Detects Early Deterioration in Acute Brain Injury
- Bacteria-Resistant Urinary Catheter Coating Reduces Infections and Antibiotic Use
- Sensor-Integrated Airway Stent Enables Continuous Remote Monitoring
- Vibration-Based Tools Aim to Improve Newborn Screening for Heart Defects
- Finger Cuff Algorithm Enables Noninvasive Screening for Aortic Stenosis
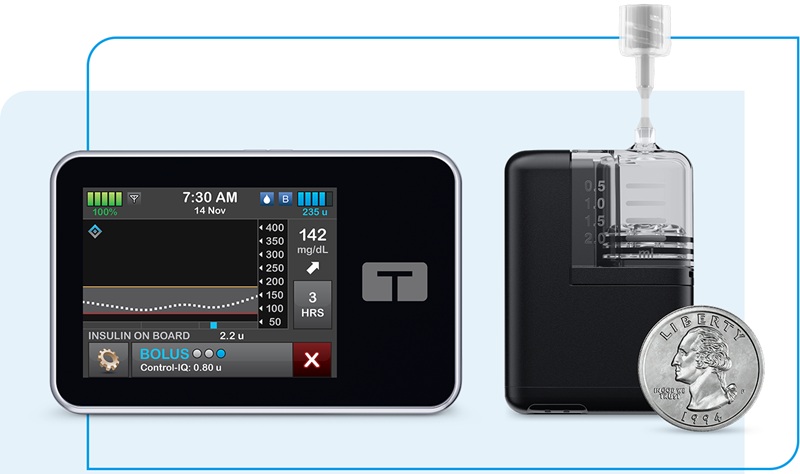
- First Automated Insulin Delivery System Cleared for Type 1 Diabetes in Pregnancy
- Novel Ablation Technology Reduces Ventricular Tachycardia Recurrence
- Dual-Energy Ablation and Conduction System Pacing Show Positive Early Outcomes
- Torqueable Microcatheters Enhance Navigation in Complex Coronary Lesions
- Transcatheter Venous Arterialization Improves Outcomes in No-Option Limb Ischemia Patients
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Proximie Advances AI-Driven Intelligent Operating Rooms with NVIDIA Collaboration
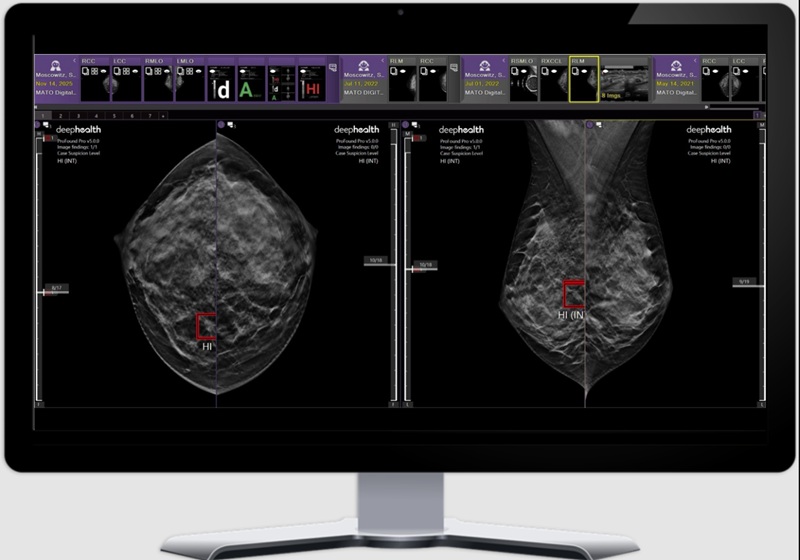
- GE HealthCare, DeepHealth Expand AI Breast Imaging Collaboration
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- New Partnership Advances Physical AI into Perioperative Workflows
- GE HealthCare and Medtronic Expand Alliance with Intraoperative Imaging Integration
- Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
- Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
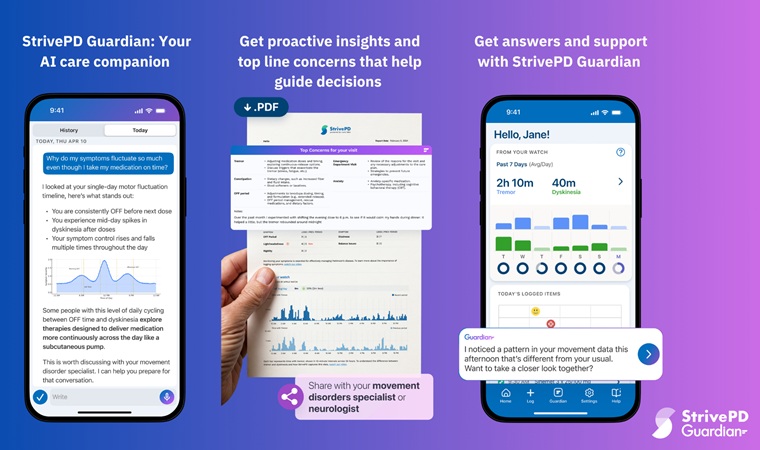
- AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data

 Expo
Expo
- Noninvasive Skull Sensor Detects Early Deterioration in Acute Brain Injury
- Bacteria-Resistant Urinary Catheter Coating Reduces Infections and Antibiotic Use
- Sensor-Integrated Airway Stent Enables Continuous Remote Monitoring
- Vibration-Based Tools Aim to Improve Newborn Screening for Heart Defects
- Finger Cuff Algorithm Enables Noninvasive Screening for Aortic Stenosis
- First Automated Insulin Delivery System Cleared for Type 1 Diabetes in Pregnancy
- Novel Ablation Technology Reduces Ventricular Tachycardia Recurrence
- Dual-Energy Ablation and Conduction System Pacing Show Positive Early Outcomes
- Torqueable Microcatheters Enhance Navigation in Complex Coronary Lesions
- Transcatheter Venous Arterialization Improves Outcomes in No-Option Limb Ischemia Patients
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Proximie Advances AI-Driven Intelligent Operating Rooms with NVIDIA Collaboration
- GE HealthCare, DeepHealth Expand AI Breast Imaging Collaboration
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- New Partnership Advances Physical AI into Perioperative Workflows
- GE HealthCare and Medtronic Expand Alliance with Intraoperative Imaging Integration
- Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
- Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
- AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data