Expo
view channel
view channel
view channel
view channel
view channel
Medical Imaging
AICritical CareSurgical TechniquesPatient CareHealth ITPoint of CareBusiness
Events
Webinars

- Automated IV Labeling Solution Improves Infusion Safety and Efficiency
- First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
- Routine Blood Pressure Readings May Identify Risk of Future Cognitive Decline
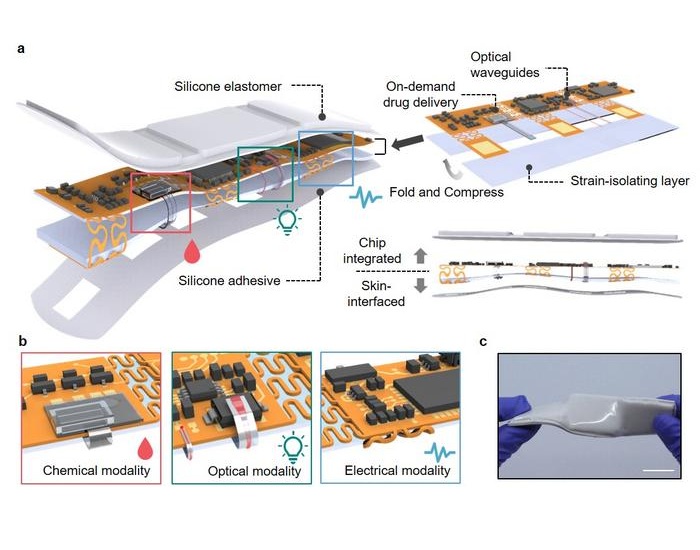
- AI Turns Glucose Data Into Actionable Insights for Diabetes Care
- 4D Digital Twin Heart Model Improves CRT Outcomes
- Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
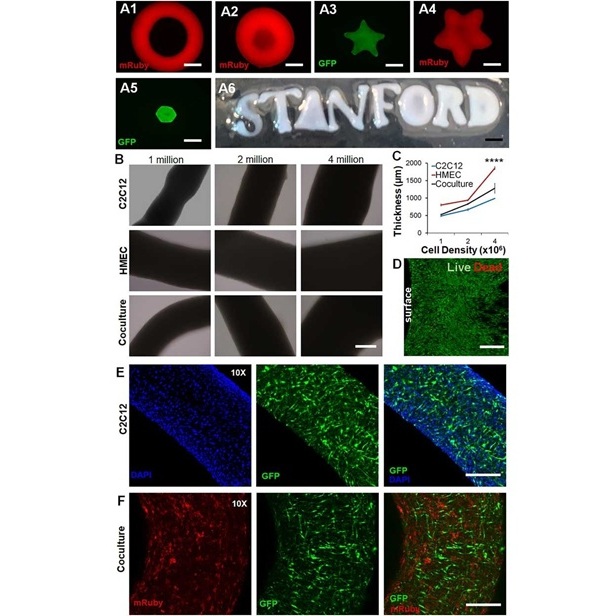
- New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
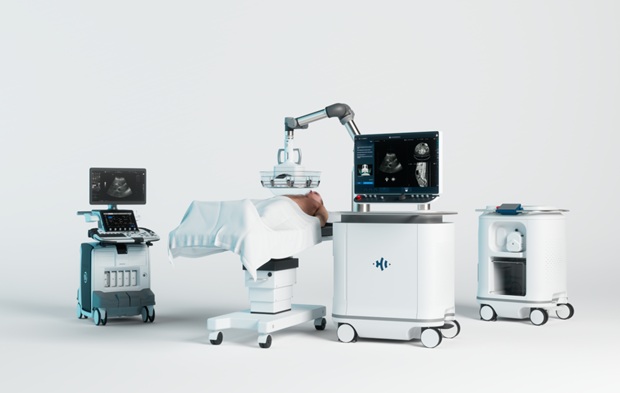
- Handheld Robotic System Expands Options for Total Knee Surgery
- VR Experience Reduces Patient Anxiety Before Kidney Stone Procedure
- Robot-Assisted Brain Angiography Improves Procedural Outcomes
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
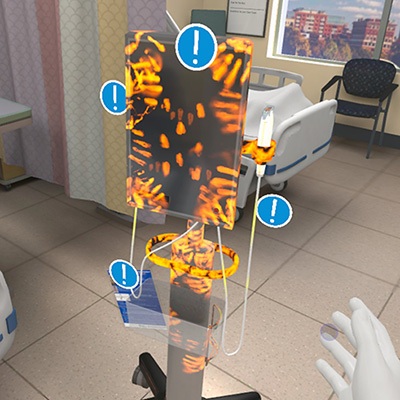
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Medtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
- Quantum Surgical Acquires NeuWave from Johnson & Johnson
- Medtronic to Acquire Coronary Artery Medtech Company CathWorks
- Medtronic and Mindray Expand Strategic Partnership to Ambulatory Surgery Centers in the U.S.
- FDA Clearance Expands Robotic Options for Minimally Invasive Heart Surgery
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
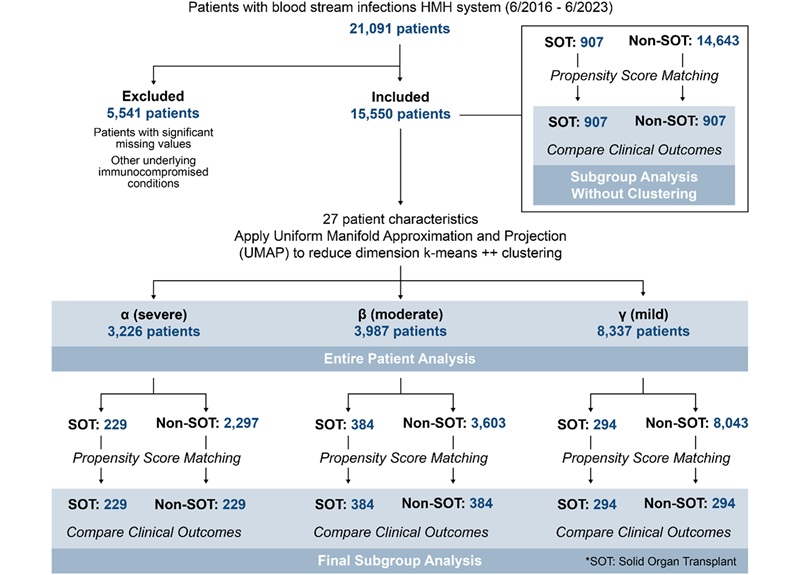
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images
- AI Tool Promises to Reduce Length of Hospital Stays and Free Up Beds

 Expo
Expo
- Automated IV Labeling Solution Improves Infusion Safety and Efficiency
- First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
- Routine Blood Pressure Readings May Identify Risk of Future Cognitive Decline
- AI Turns Glucose Data Into Actionable Insights for Diabetes Care
- 4D Digital Twin Heart Model Improves CRT Outcomes
- Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
- New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
- Handheld Robotic System Expands Options for Total Knee Surgery
- VR Experience Reduces Patient Anxiety Before Kidney Stone Procedure
- Robot-Assisted Brain Angiography Improves Procedural Outcomes
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Medtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
- Quantum Surgical Acquires NeuWave from Johnson & Johnson
- Medtronic to Acquire Coronary Artery Medtech Company CathWorks
- Medtronic and Mindray Expand Strategic Partnership to Ambulatory Surgery Centers in the U.S.
- FDA Clearance Expands Robotic Options for Minimally Invasive Heart Surgery
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images
- AI Tool Promises to Reduce Length of Hospital Stays and Free Up Beds