Expo
view channel
view channel
view channel
view channel
view channel
view channel
Medical Imaging
AICritical Care
Patient CareHealth ITPoint of CareBusiness
Events
Webinars

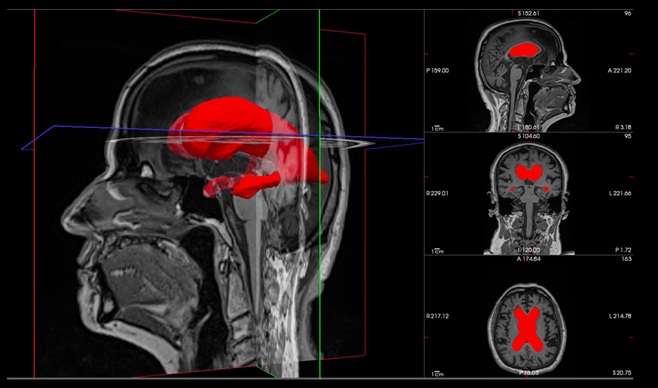
- 3D Brain Imaging Marker Predicts Surgical Outcomes in Dementia-Causing Hydrocephalus
- Printed Artificial Neurons Stimulate Natural Brain Circuits
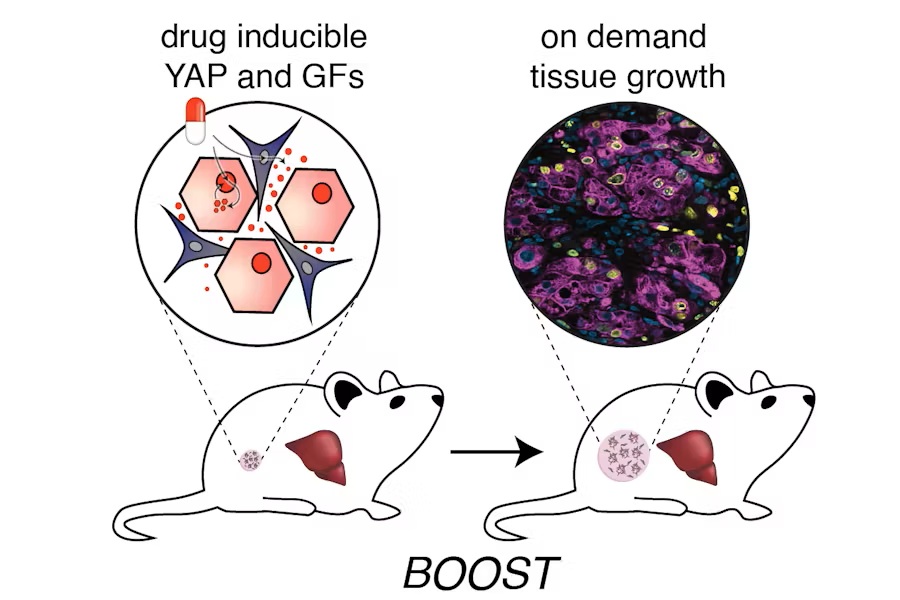
- Synthetic Biology Approach Enables On-Demand Liver Tissue Growth
- Bioinspired Imaging System Identifies Cancerous Lymph Nodes Intraoperatively
- FDA-Cleared ECG Software Powers Real-Time Cardiac Monitoring at Scale
- Innovative Microsurgery Advances Treatment of Cancer-Related Lymphedema
- Bioengineered Ligament Aims to Improve ACL Repair Outcomes
- Thrombectomy Device with Real-Time Control Targets LVO Stroke
- Fish-Skin Graft Shortens Hospital Stay in Severe Burns
- Transcatheter Valve Replacement Demonstrates High Success in Real-World Study
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- New Partnership Advances Physical AI into Perioperative Workflows
- GE HealthCare and Medtronic Expand Alliance with Intraoperative Imaging Integration
- External Liver Assist System Receives FDA RMAT Designation
- New Partnership Expands Access to Predictive Tool for Patient Monitoring
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
- Smartwatches Could Detect Congestive Heart Failure
- Versatile Smart Patch Combines Health Monitoring and Drug Delivery
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections

 Expo
Expo
- 3D Brain Imaging Marker Predicts Surgical Outcomes in Dementia-Causing Hydrocephalus
- Printed Artificial Neurons Stimulate Natural Brain Circuits
- Synthetic Biology Approach Enables On-Demand Liver Tissue Growth
- Bioinspired Imaging System Identifies Cancerous Lymph Nodes Intraoperatively
- FDA-Cleared ECG Software Powers Real-Time Cardiac Monitoring at Scale
- Innovative Microsurgery Advances Treatment of Cancer-Related Lymphedema
- Bioengineered Ligament Aims to Improve ACL Repair Outcomes
- Thrombectomy Device with Real-Time Control Targets LVO Stroke
- Fish-Skin Graft Shortens Hospital Stay in Severe Burns
- Transcatheter Valve Replacement Demonstrates High Success in Real-World Study
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- New Partnership Advances Physical AI into Perioperative Workflows
- GE HealthCare and Medtronic Expand Alliance with Intraoperative Imaging Integration
- External Liver Assist System Receives FDA RMAT Designation
- New Partnership Expands Access to Predictive Tool for Patient Monitoring
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
- Smartwatches Could Detect Congestive Heart Failure
- Versatile Smart Patch Combines Health Monitoring and Drug Delivery
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections