Expo
view channel
view channel
view channel
view channel
view channel
view channel
Medical Imaging
AI
Surgical TechniquesPatient CareHealth ITPoint of CareBusiness
Events
Webinars

- New Technology Turns Earbuds into Sensors for Cardiac Function Tracking
- Wearable AI Tool Estimates Vascular Age for Cardiovascular Risk
- New Brain Stimulation Approach Targets Deep Brain Areas Without Surgery
- Standardized FMT Protocol May Improve Survival in Severe C. difficile Infection
- Injectable Microgel Reduces Blood Loss in Infant Surgery
- Dynamic Pressure Overlay Reduces Perioperative Pressure Injuries
- Pulsed Field Ablation System Streamlines Atrial Fibrillation Procedures
- Single-Use System Enables Minimally Invasive Decompression for Lumbar Spinal Stenosis
- Angiography-Based Tool Matches Standard FFR for Coronary Revascularization Guidance
- Endoscope Enables Fallopian Tube Imaging and Cell Collection for Ovarian Cancer Surveillance
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
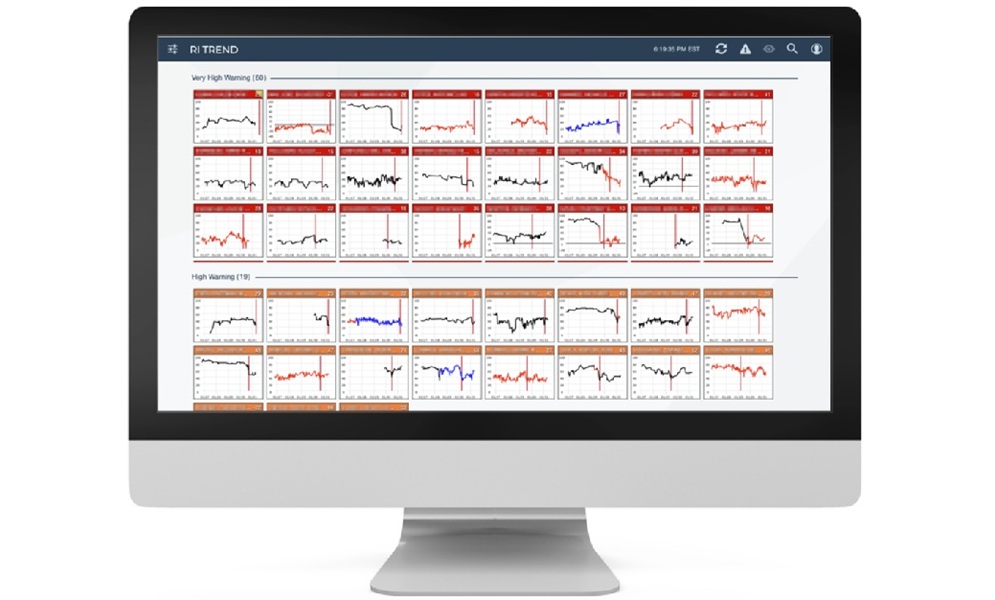
- New Partnership Expands Access to Predictive Tool for Patient Monitoring
- GE HealthCare Leads Major European Initiative to Advance Cardio-Oncology Care
- Medtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
- Quantum Surgical Acquires NeuWave from Johnson & Johnson
- Medtronic to Acquire Coronary Artery Medtech Company CathWorks
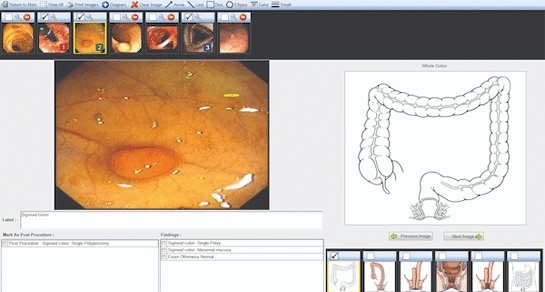
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
- Smartwatches Could Detect Congestive Heart Failure
- Versatile Smart Patch Combines Health Monitoring and Drug Delivery
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images

 Expo
Expo
- New Technology Turns Earbuds into Sensors for Cardiac Function Tracking
- Wearable AI Tool Estimates Vascular Age for Cardiovascular Risk
- New Brain Stimulation Approach Targets Deep Brain Areas Without Surgery
- Standardized FMT Protocol May Improve Survival in Severe C. difficile Infection
- Injectable Microgel Reduces Blood Loss in Infant Surgery
- Dynamic Pressure Overlay Reduces Perioperative Pressure Injuries
- Pulsed Field Ablation System Streamlines Atrial Fibrillation Procedures
- Single-Use System Enables Minimally Invasive Decompression for Lumbar Spinal Stenosis
- Angiography-Based Tool Matches Standard FFR for Coronary Revascularization Guidance
- Endoscope Enables Fallopian Tube Imaging and Cell Collection for Ovarian Cancer Surveillance
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
- New Partnership Expands Access to Predictive Tool for Patient Monitoring
- GE HealthCare Leads Major European Initiative to Advance Cardio-Oncology Care
- Medtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
- Quantum Surgical Acquires NeuWave from Johnson & Johnson
- Medtronic to Acquire Coronary Artery Medtech Company CathWorks
- Voice-Driven AI System Enables Structured GI Procedure Documentation
- EMR-Based Tool Predicts Graft Failure After Kidney Transplant
- Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
- Smartwatches Could Detect Congestive Heart Failure
- Versatile Smart Patch Combines Health Monitoring and Drug Delivery
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images